Autism Spectrum Disorder (ASD) is a neurological condition affecting 1 in 59 children in the United States. ASD can be described using four main developmental and behavioral categories: social skills, repetitive behaviors, speech, and nonverbal communication. Every child exhibits different strengths and abnormalities in each category.
There is no cure for ASD. We also do not know exactly what causes it. Researchers are actively investigating causes and treatment options. The ultimate goal is to develop resources for children with ASD and their families to offer the best life for them.
More Information: CDC Resource on Autism
A limited number of evidence-based therapies exist at this time. Motivated parents have started looking at alternative therapies to support the growth and development of their children. But, without strong evidence, clinicians have not established the safety or efficacy of these therapies.
In this article, we examine the safety of stem cell therapy as a treatment for ASD.
Study Design
The study measured the safety of stem cell therapy in ASD patients. Children with a confirmed diagnosis of ASD and a qualified banked autologous umbilical cord blood unit were eligible to participate.
The sample included 25 children, mostly white males, between the ages of 2 and 5. The majority of children were categorized as having moderately severe or severe ASD symptoms.
Children received their stem cell infusion under supervised care and were tracked for a year. The children went to check-ups at 6 and 12 months after the infusion so a doctor could assess their health. Their caregivers filled out detailed surveys every three months after the infusion.
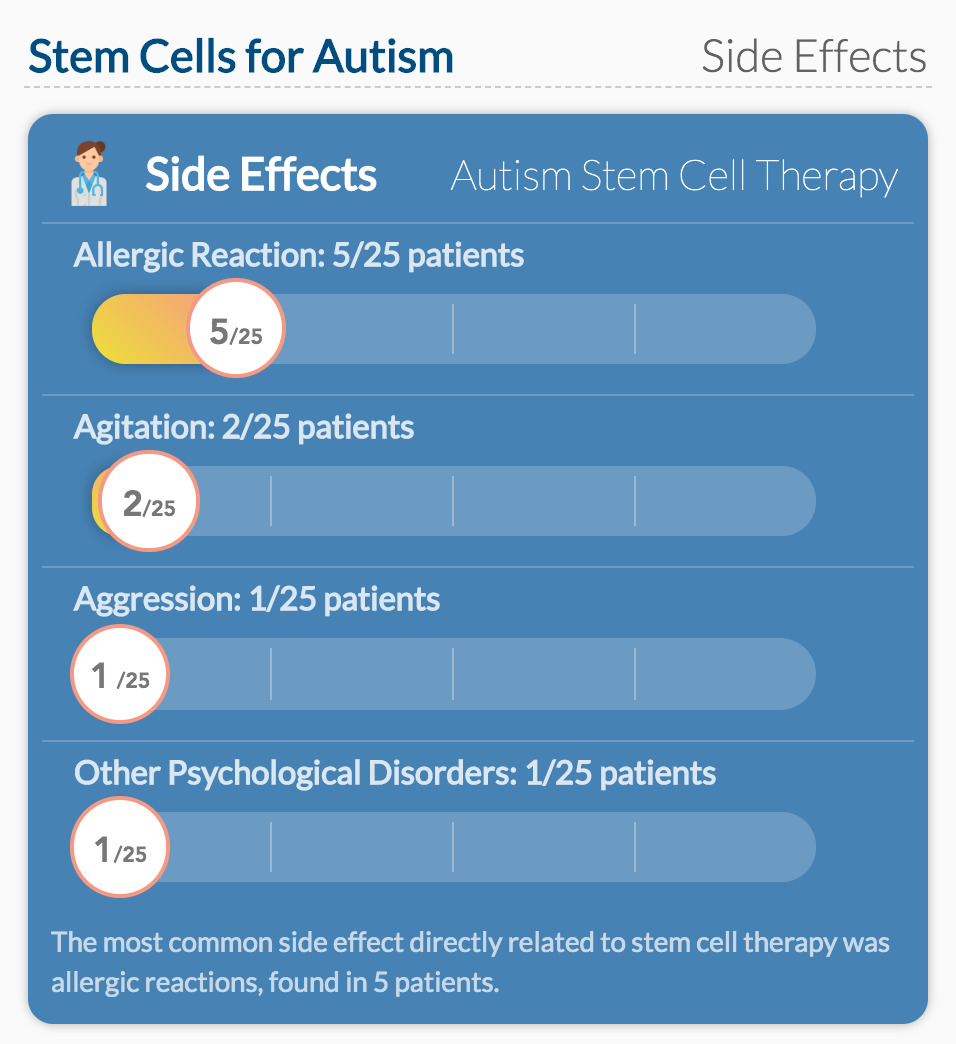
Side effects
The good news is that there were no serious side effects reported in any of the children. Of all the side effects reported, only 13% of them were considered to be related to stem cell therapy. Five children had an allergic reaction to the therapy, causing a skin rash or cough. These events occurred on the day of infusion and resolved quickly with a dose of Benadryl.
Cases of agitation, infections, and other medical events were deemed unrelated to the infusion. Agitation, in particular, was considered to be a common symptom of ASD and not caused by any treatment.
Behavioral change
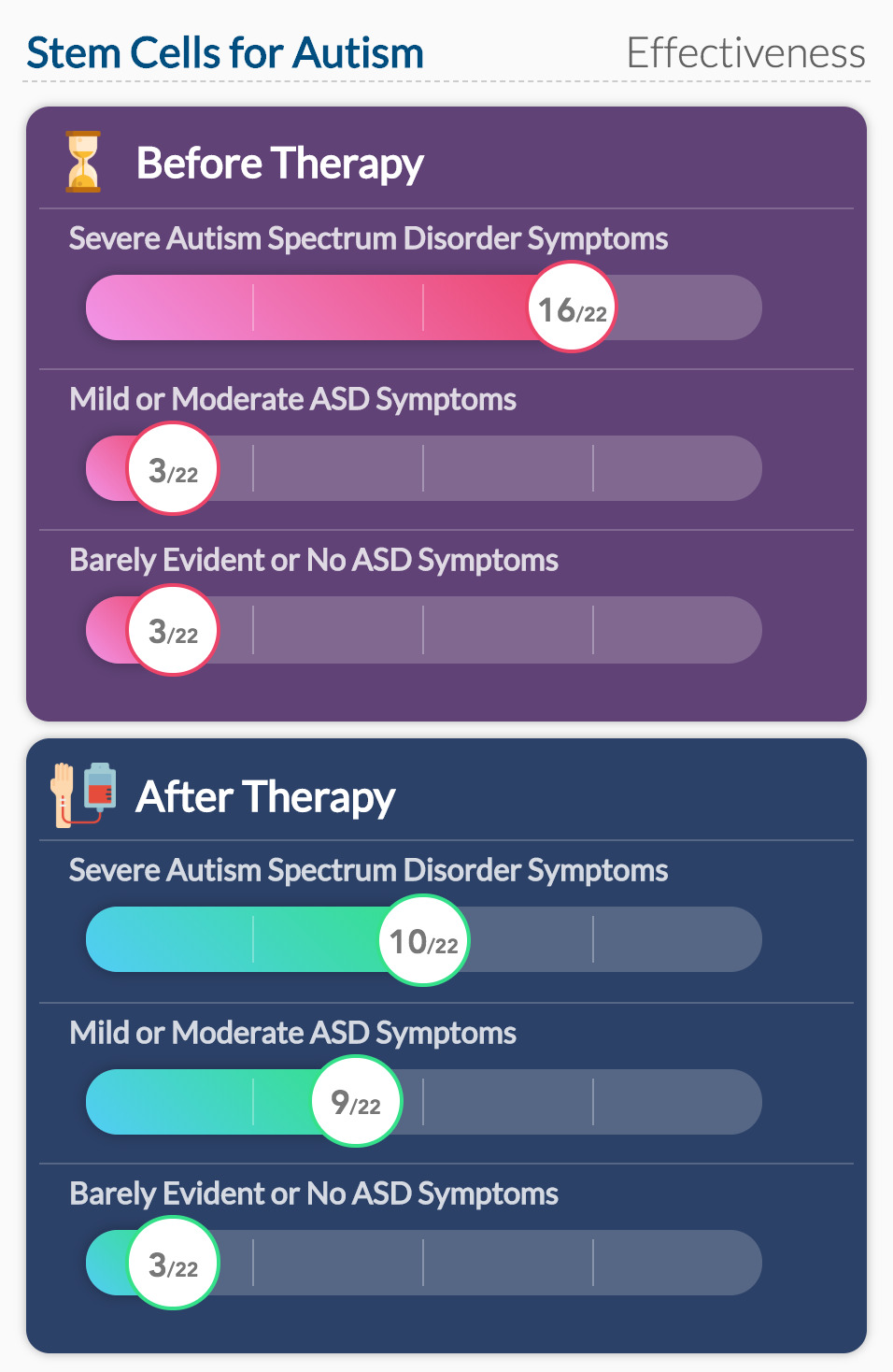
Along with measuring the safety of stem cell therapy, this study also collected data on behavioral changes during the year follow-up. Researchers used the Clinical Global Impression (CGI) scale to measure changes in symptom severity over time.
Six months after infusion, the number of participants with moderately severe and severe symptoms decreased. Adaptive behaviors and socialization reportedly improved.
What this study really tells us
Over the year of follow-up, all health concerns among participants were considered mild, expected, and easily resolved. The majority of side effects were found to be unrelated to the infusion itself and generally common among children.
Before we draw any bold conclusions, we must consider a few things. First, the sample size was very small. Even though no severe adverse events occurred in this sample, that does not mean they won’t happen in a larger population. Second, most of the participants were white males, so these findings can not speak for other demographics. Lastly, the study had no placebo or control group, meaning there was no group to compare with.
As for the behavioral changes, it is not possible to directly associate stem cell therapy to ASD symptom improvement. These children were very young, so change is normal and accelerated at that time in their life. The symptom improvement could have been due to other behavioral therapies. In summary, the study did not consider all other possible reasons that the children’s ASD symptoms improved.